Butterfly Needle Uses, Advantages, and Limitations
Butterfly Needle Applications
The butterfly needle is one of the most common, versatile, and popular medical devices used across various medical and healthcare professions. Here, we’ll take a closer look at the butterfly needle and explore its many uses, technical advances, and considerations in the routine use of these devices.
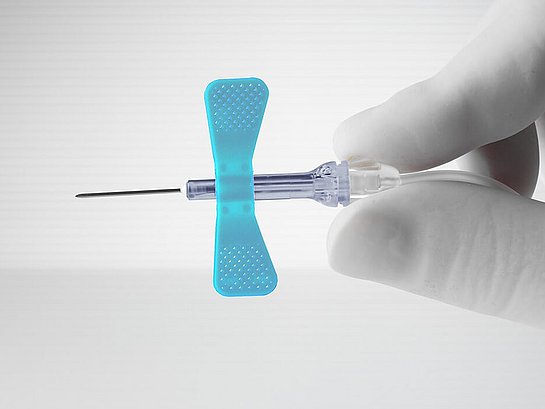
The butterfly needle is one of the most common, versatile, and popular medical devices used across various medical and healthcare professions. Butterfly needles are uniquely designed devices that are used for drawing blood. Some butterfly needles can also be used for infusion of medications intravenously. The term “butterfly needle” comes from the unique wing-like designs that flank the needle itself. Here, we’ll take a closer look at the butterfly needle and explore its many uses, technical advances, and considerations in the routine use of these devices.
It’s possible to find butterfly needles in workplaces across the medical spectrum. Doctors, nurses, medical researchers, laboratorians, and, of course, phlebotomists all make use of butterfly needles on a regular basis. Butterfly needles are most often associated with the practice of phlebotomy –– typically, the withdrawal of a blood sample for the purpose of laboratory testing.
In addition, they can also be used to transfer medications intravenously –– particularly in the pediatric population or when a patient cannot take medicine orally. Butterfly needles are available with options in tubing length, needle gauge and adapters for a variety of applications.
Butterfly Needle Benefits & Advantages
Butterfly needles are one of the most widely used types of needle by hospital staff. There are several reasons why this is the case. First, the design of the butterfly needle allows for easy manipulation. Many healthcare workers find it easier to use a butterfly needle to draw blood than other devices. Second, because healthcare workers feel more comfortable with butterfly needles –– and because butterfly needles are available with smaller gauges –– they are considered a good option for drawing blood from patients with small or hard-to-access veins. Third, butterfly needles are less likely to cause[1] [2] nerve injury and similar complications given their shorter length relative to other needle types, according to this report.
In summation, butterfly needles are often favored by both healthcare workers and patients.
Butterfly Needles & Needlestick Injuries
Though butterfly needles are popular, there are a few potential considerations in their routine use. Like any needle, butterfly needles carry the risk of percutaneous injury. The Centers for Disease Control and Prevention (CDC) estimates that each year 385,000 needlestick injuries (NSIs) and other sharps-related injuries are sustained by hospital-based healthcare personnel. Unfortunately, the risks and costs associated with NSIs and blood exposure are serious and real. There are direct costs associated with the care and treatment for healthcare personnel that sustain injuries and there are indirect costs associated with the emotional toll of possible exposure. One study1 conducted in two hospitals observed that healthcare personnel who reported a sharps-related injury were willing to pay a median amount of $850 in order to avoid a sharps injury; this amount increased to over $1000 when adjusted for patient risk status and working with an uncooperative patient at the time of injury. Study investigators concluded that in order to avoid the anxiety and distress induced by NSIs, healthcare personnel were willing to pay amounts similar to the costs of post-exposure evaluation. Therefore, there is certainly a financial and emotional burden to consider with any needle-containing device.
With that being said, the added safety components of newer butterfly needles have significantly reduced the possibility of needlestick injuries for healthcare workers. Butterfly needles, like the VACUETTE® EVOPROTECT, are now equipped with mechanisms that provide semi-automatic retraction of the needle from the vein, which minimizes the risk of needlestick injury.
According to a study2 based in France, safety-engineered devices with a semi-automatic safety feature are more effective at preventing NSIs than devices with a manually activated mechanisms. Researchers noted semi-automatic safety devices were involved in 1.18 NSIs per 100,000 purchased. In contrast, devices with a manually activated toppling shield accounted for 2.94 NSIs per 100,000 purchased, and devices with a manually activated sliding shield were involved in 5.20 NSIs per 100,000.
Many of the incidents documented in the study occurred during activation of the safety feature or were attributed to failure to appropriately activate it. The authors concluded that devices with semi-automatic features were particularly effective at preventing NSIs during activation. Semi-automatic devices help to lower the risk during these phases of application accounting for just 3 NSIs during activation in the course of the study research. Manually activated devices were associated with significantly more injuries in part because they rely on the user to adhere to manufacturer instructions for proper activation.
Though the introduction of safety engineered devices has had a considerable impact on the occurrence of NSIs, products like GBO’s VACUETTE® EVOPROTECT that utilize a semi-automatic safety feature reduce the risk of injuries associated with winged devices even further.
The Greiner Bio- One Difference
At Greiner Bio-One the principles of safety and accuracy direct everything we do. We offer a wide range of highly sophisticated, safe, and reliable butterfly needles that can be used for a wide variety of applications. If you’d like to learn more about our products, you can contact one of our team members today.
- Fisman DN, Mittleman MA, Sorock GS, Harris AD. Willingness to pay to avoid sharps-related injuries: a study in injured health care workers. Am J Infect Control 2002; 30(5):283- 7.
- Tosini MD, Ciotti RN, Goyer, RN, Lolom MSc, L’He´riteau MD, Abiteboul MD, Pellissier PhD, Bouvet MD. Needlestick Injury Rates According to Different Types of Safety-Engineered Devices: Results of a French Multicenter Study. Injection Control and Hospital Epidemiology 2002; 31(5).